|
1/2/2023 0 Comments Fda iso 13485
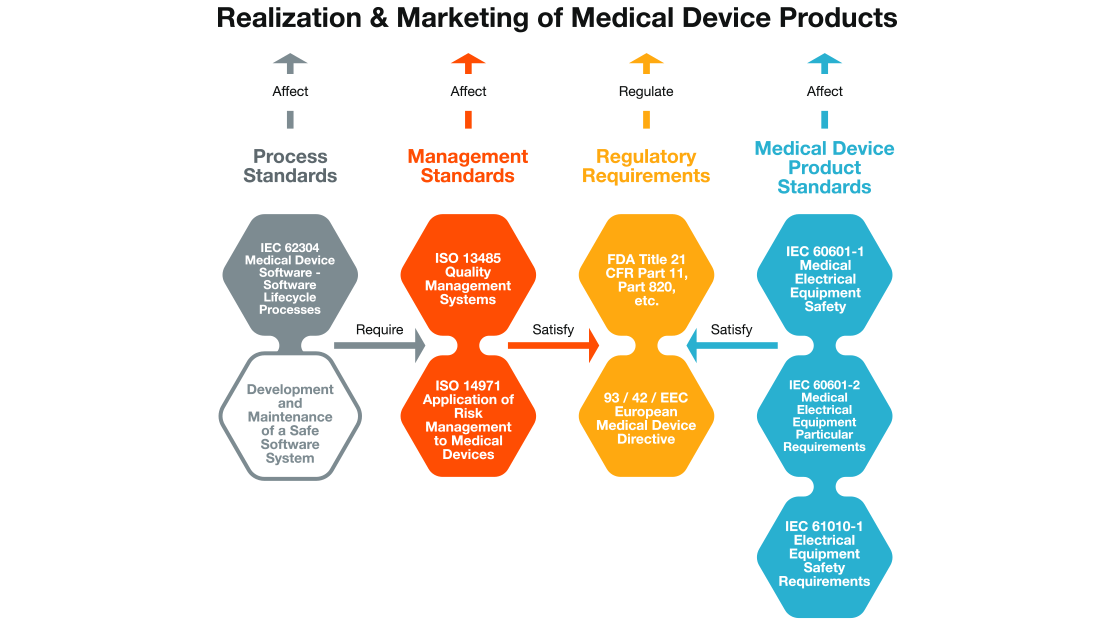
The technical committee said the definitions of risk found in the HLS does not align with regulatory definitions, but there is also some concern over whether the description of risk in the HLS is sufficiently prescriptive to be enforceable in the courts. One of the tensions in incorporating the HLS language into 13485 is the way the term “risk” is defined. The Medical Device Single Audit Program (MDSAP) by the International Medical Device Regulators Forum (IMDRF) also relies on 13485, making this a critical piece of the regulatory harmonization picture, and thus nearly impossible to set aside. The implementation date for the MDRs has been delayed to May 26, 2021, although the EU had appended several annexes to the standard. 
For reasons that are not explained, removing 13485 from the list of standards that are subject to the HLS requirement received little attention in the committee’s statement.Ī fourth option for easing the HLS problem would be to “forget about ISO altogether,” the committee said, an option that may seem unlikely, given that the European Union has adopted 13485 for its Medical Device Regulations (MDRs). The proposal to modify HLS was seen as the second most plausible option, although removal of 13485 from the list of HLS-mandated standards was also under consideration.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
